The China National Intellectual Property Administration ("CNIPA") recently announced the "Revised Patent Examination Guidelines", which came into force from January 15, 2021. This is a major development in the field of IP industry in China. Although the "Revised Guideline" is not a formal source of law, it can be used as a reference in courts' decisions provided that there are no conflicts with the "Chinese Patent Law" and its implementation rules. Therefore, its amendment is of great significance to the administrative and judicial procedures on patent-related matters.
In this article, we just focus on main amendments addressed in the "Revised Guideline".
Supplementary Experiment Data Submission
In the past, both CNIPA and the court took a relatively strict position against the post-filing supplementary data. As a result, it was generally more difficult to obtain a granted pharmaceutical patent in China than in other jurisdictions. However, under the pressure of "Economic and Trade Agreement Between China and U.S." ("Agreement") where "China should allow pharmaceutical patent applicants to submit supplementary data in patent examination procedures, reexamination proceedings and judicial proceedings to meet the relevant requirements of patentability, including the requirements for sufficient disclosure and inventive steps", the CNIPA decides to change the game rule.
The "Revised Guidelines" provides at least two examples under this topic:
[Example 1] Compound A is claimed. The specification describes a method of preparing Compound A, and its effect in reducing the blood pressure, and an experimental method for measuring the activity of reduced blood pressure, but the experimental result data is not disclosed. In order to prove that the specification fully disclosed the invention, the applicant supplemented the data to show the effect of Compound A in reducing the blood pressure. For those skilled in the art, according to the original application documents, the effect of reducing the blood pressure of Compound A has been disclosed, and thus the technical effect shown in post-filing supplementary data is derivable from the disclosure of the patent application, and thus the supplementary data should be considered and examined.
[Example 2] A compound of general Formula I is claimed. The specification describes the general Formula I, a method of preparing Formula I, and specific compounds A, B, and the like. The specification also describes the antitumor effect of Formula I and the experimental methods and experimental data of determining the antitumor activity. The data shows that IC50 value is between 10-100nM. The Applicant submitted supplementary experiment data to show the inventive steps, which shows that IC50 value of Compound A is 15nM and the compound in reference 1 is 87nM. For those skilled in the art, according to the original application documents as filed, compound A and its antitumor effects have been disclosed, and thus the technical effects shown in the postfiling supplementary data is derivable from the disclosure of the patent application, and thus the supplementary data should be considered and examined.
Use Limitation of Composition Claims
According to the old Patent Examination Guideline, when the composition has two or more uses and properties, the use does not have to be recited in the claims; however, when only one use or one property is disclosed, the composition claim should be limited by its use.
The "Revised Guidelines" provides a possibility to draft the composition claim without any use limitation even if the specification only discloses one use or one property of the composition.
Novelty of Compounds
The "Revised Guidelines" states: "where a patent application claims a compound, if the chemical name, molecular formula (or structural formula) and other structural information of the compound is recorded in a reference so that those skilled in the art believe that the claimed compound has been disclosed, the compound is not novel, unless the applicant can provide evidence to prove that the compound cannot be obtained before the filing date." In other words, even if the reference has disclosed the same physical and chemical parameters as the claimed compound, it cannot be directly inferred that the compound is not novel.
In addition, according to the "Revised Guidelines", when the structural similarities and differences between the claimed compounds and reference cannot be determined, physical and chemical parameters, preparation and experimental effects, and other factors should be combined and considered to determine whether the compounds can be predicted as substantially the same by the skilled in the art. On the contrary, the applicant can prove the novelty of the compound by proving the structural difference between the claimed compound and the reference.
Inventive Steps of Compounds
The "Revised Guidelines" has made major changes to the examination of inventive steps of compounds, that is, regardless of whether the compound is similar in structure to the existing compound, a unified "problem-solutionapproach" is used to determine whether a compound is inventive.
Two examples provided by the "Revised Guidelines" are indicated below:
[Example 1]
Prior art:
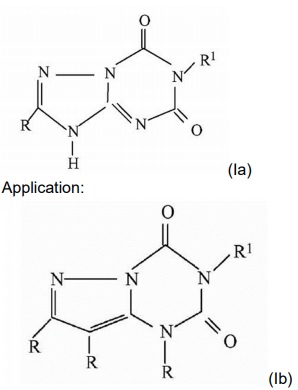
(Ib) and (Ia) have different core structures, but they have the same use. Those skilled in the art generally believe that compounds with similar structures have the same or similar uses, and similar structures generally mean that the compounds have the same basic core part or basic ring. In the prior art, there is no technical inspiration about modifying the basic ring of (Ia) to obtain (Ib) while maintaining the same use, therefore, (Ib) meets the requirement of inventive steps.
[Example 2]
Prior art: H2N-C6H4-SO2NHR1 (IIa)
Application: H2N-C6H4-SO2-NHCONHR1 (IIb)
(IIb) is obtained by inserting -CONH- into the structural fragment NHR1 of (IIa). (IIa) and (IIb) have different uses as (IIa) is a sulfa antibiotic while (IIb) is sulfonyl acyl urea and an anti-diabetic drug. Those skilled in the art have no motivation to modify R1 in the sulfa antibiotic into CONHR1 to obtain the anti-diabetic drugs. Therefore, (Ib) meets the requirement of inventive steps.
Deposition
Guangdong Culture Collection Center (GDMCC) located in Guangzhou is added.
Monoclonal Antibodies
Regarding the drafting of monoclonal antibody claims, the "Revised Guidelines" adds a limitation method of "limiting by structural features".
It is stipulated in the current Guidelines that monoclonal antibody claims can be defined by the hybridoma from which it is produced. However, with the maturity and popularization of monoclonal antibody sequencing technology, it has become easier to obtain structural information of monoclonal antibodies and it is possible to define monoclonal antibodies by structural features.
The "Revised Guidelines" provides the following examples under this topic:
[Example]
- A monoclonal antibody of antigen A, comprising VHCDR1, VHCDR2, and VHCDR3 shown in amino acid sequences of SEQ ID NOs. 1-3, and VLCDR1 VHCDR2, and VHCDR3 shown in amino acid sequences of SEQ ID NOs. 4-6.
- A monoclonal antibody of antigen A produced by hybridoma of CGMCC Deposition No: XXXX.
Inventive Steps in Biotechnology
Regarding the inventive steps of biotechnology, the "Revised Guidelines" discarded the over-absolute requirement of "unexpected technical effect" and returned to the "problem-solution-approach" for examination. In addition, the "Revised Guideline" also enriched subject matters and added new forms of biotechnology such as "peptides or proteins".
The content of this article is intended to provide a general guide to the subject matter. Specialist advice should be sought about your specific circumstances.



